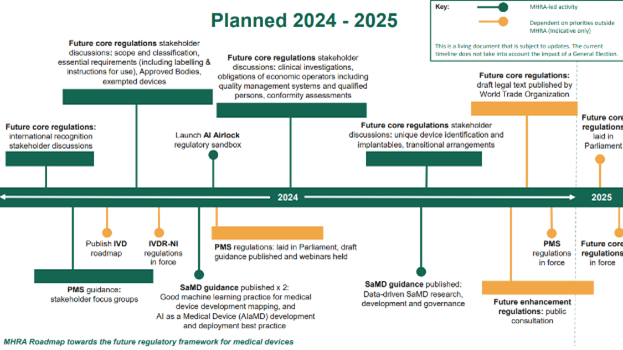
A new MedTech roadmap published by the MHRA The MHRA have today published a roadmap towards the future regulatory framework for medical devices. This sets out their planned activities into 2025. The roadmap is intended to deliver greater international harmonisation, with more patient-centred, proportionate requirements for medical devices which are responsive to technological advances. Dr Laura Squire, the MHRA’s MedTech Regulatory Reform…
DawnJanuary 10, 2024