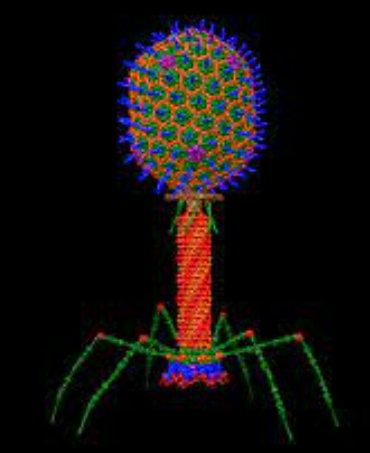
Proposal start date: 11/07/2023 Proposal end date: 02/09/2023 Scope This document describes the minimum requirements for bacteriophage preparation processing including the assessment of the titer and quality control. This document applies to data processing of bacteriophage isolation, culture, purification and storage. This document applies to the quality evaluation/assessment of bacteriophage used for therapy. Purpose With the continuous and expanded…
Ben KempSeptember 8, 2023