Qserve perspective on the new Australian TGA UDI requirements. Increasingly relevant as Australia is one of the countries MHRA have identified from international recognition under the proposals for the Pre-Market SI.
*Please note, the above post on TGA UDI requirements shows the timelines table for medical devices rather than the IVD table as below.
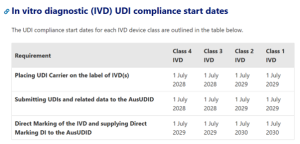
TGA UDI Timelines for IVDs
complying-unique-device-identification-requirements-medical-devices.pdf
In vitro diagnostic devices in scope of UDI requirements
The following table describes which IVD classes must meet UDI requirements. These devices must meet all UDI requirements, unless specifically exempt.
IVD classification UDI required?
Class 1 Partial*
Class 2 Yes
Class 3 Yes
Class 4 Yes
*IVDs that are instruments and software are required to comply with the UDI requirements. Such devices are typically covered under the following GMDN https://www.gmdnagency.org/ Term:
- instrument/analyser IVDs (GMDN Code CT943)
- software IVDs (GMDN Code CT944).
IVDs that are also software may have further requirements. See medical device and IVD software section of the guidance.
As a manufacturer, you may choose to assign and apply a UDI to other exempt IVDs. If you do apply a UDI to these devices, we recommend you meet all UDI requirements to reduce confusion for end users such as healthcare.
As a sponsor, you may choose to submit and maintain the relevant data to the AusUDID for other exempt IVDs. If you do submit the data for these devices to the AusUDID, your data must comply with UDI requirements. Note that your device must have an applicable ARTG inclusion to make the data available in the AusUDID.
Devices exempt from UDI requirements
You are not required to meet UDI requirements for:
medical devices that are Class I non measuring nonsterile
medical devices that are Class I measuring (Im)
in house IVDs
custom-made medical devices
patient-matched medical devices with a volume of 5 or less supplied each financial year
medical devices exempt under Special Access Scheme (SAS) or Authorised Prescriber (AP) Scheme
Surgical Loan Kits (SLKs) at kit level.
While these devices are exempt from complying with UDI requirements, you may choose to meet UDI requirements for these devices.
