
NHS England and NICE have launched a consultation on a proposed integrated, rules-based pathway for the evaluation, funding, and commissioning of medical technologies.
It’s been recognised after years of system-wide feedback that the current process is challenging for our established members and even more so for new entrants. It also demonstrates their understanding around a marked difference in approach between the assessment of medicines and MedTech.
The proposal sets out the rules and principles within which NICE and NHS England will work in partnership to evaluate, commission and fund medical technologies.
It outlines how the NHS will support the routine commissioning of technologies determined to be clinically and cost effective by NICE in the NHS.
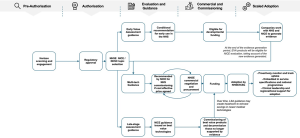
The are 5 key areas included:
- Evidence-based advice and guidance from the National Institute for Health and Care Excellence (NICE), focused on technologies with the greatest impact on patient outcomes and the most compelling cases for clinical and cost-effectiveness.
- A lifecycle approach to new, early-stage technologies and driving greater value from existing technologies in widespread use.
- Automatic identification of funding to support commissioning and adoption of clinically cost-effective and affordable technologies.
- Support the transformation of clinical pathways and services.
- Drive up the quality and use of evidence, helping tackle ethnic disparities and unfair biases in MedTech.
System-wide integration, clarity and consistency should support industry by offering a better understanding of process and demand-signalling. The proposals also nod to the recently announced MHRA regulatory roadmap by prioritising those products with the greatest impact.
On the face of it, this might deliver better engagement between manufacturers, suppliers and the NHS. It should enable better horizon-scanning and responsiveness and has the potential to support automatic funding solutions for appropriate devices, providing it is done well.
The NHS has historically failed to understand that it is part of a global market and the economies of scale and buyer power it considers it holds isn’t as significant as it might once have thought. It is welcome that this realisation of how industry works globally is starting to be clearer. BIVDA supports the overall principle of addressing the approach to MedTech and medicines and the ambition to introduce a national set of rules for recommendation and reimbursement.
However, what this consultation does not adequately cover is the translational piece between policy and implementation, policy and practice and policy and acceptance. There is still the elephant in the room relating to the relationship between the central healthcare functions who do “get it” and want to deliver a change to the system, and the system itself, where clinical preference, cost improvement requirements, dysfunctional connectivity between acute and community settings, workforce pressures and resistance to clinical pathway change remains a challenge.
No individual within the NHS is fundamentally resistant to any MedTech innovation that improves patient experience or relieves workforce pressures. It is the system itself which isn’t geared up for it, and this is an area that the consultation doesn’t cover in any depth. It recognises the area needs addressing, which is progress, but the key to whether the rules-based pathway does what it aims to is in our view wholly dependent on whether it is implemented well and accepted by those expected to deliver.
The detail around commissioning, the incentives available, the rewards and sacrifices required across all the elements, is where we need to see effort applied. Otherwise, it will sit somewhere between guidance and a wish list due to the pressures of delivering the existing requirements – just one more thing to consider at a later date or when there is time, or when the risk to service is lower or after someone else goes first.
Alongside the need to understand the detail around translation, two other points pique our interest.
- a reliance on the capacity and capability of NICE to deliver.
- no (equivalent) national funding to level MedTech and medicines. NHSE and NICE clearly recognise this – and their control measure is to initially limit the proposed scope to five products per year before scaling up.
It’s no secret that there is sector-wide concern about what “value assessment” means in reality. The proposal suggests “NHS England’s commercial function will communicate with industry on how the NHS perceives the ‘value’ of MedTech, considering feasibility and implementation alongside clinical outcomes.” But this must be called out. This requires real engagement and a real understanding, in addition to resources and skills, not just lip service, or desktop consultations.
Historically, this has only really been one way and based on the principle of engaging once the decision is made. In practice, this is effectively negotiation and applied with a commercial lens.
Engagement and value must be addressed in a non-competitive way, allowing market forces to drive competition at the point of procurement or commissioning. Industry (this means Trade Associations and suppliers) must be completely equal and reach mutual agreement in these conversations, and the collaboration true.
Trust between industry and the NHS needs to repair to address long term behaviours which are embedded into NHS activities through years of policy, procedures and guidance which need to be unravelled. There is better engagement and consultation in relationships between industry and the central functions, but this needs to filter into plans around implementation and while this consultation acknowledges the areas to address it doesn’t go into how. In principle, as it’s written, this rules-based pathway could work. It’s clear the intent and will is there, but it’s difficult to make a call without understanding how the proposal will be rolled out, embedded and measured.
BIVDA are cautiously optimistic regarding the direction of travel, this is an example of the better understanding of the innovation landscape challenges, and it’s clear that those involved have been listening to feedback from industry. However, there needs to be a commitment to work with industry to find a path to implement and scale this with explicit involvement of industry to define value and to embed a process where innovation is funded for IVD products and processes.
BIVDA has been consulted along with other Trade Associations in the MedTech space alongside this public consultation, and whilst in principle this is absolutely a step in the right direction for the sector as a whole, for IVDs it is still not quite right in its current form. It doesn’t address managed services, which is part of the Laboratory consolidation maturity requirements within NHS transformation and the relationships manufacturers have with Primary contractors, nor long term contracts of ten years plus and the regulations that govern them.
This must be the start of a continued collaborative effort to understand the IVD anomalies and touch points of the proposal throughout the system and deliver better adoption of technology across the Sector.
BIVDA will collate members views as part of our response to the consultation and encourage you to respond directly. Please provide your responses to us for inclusion by emailing ben.kemp@bivda.org.uk.
The consultation response and BIVDA activity will be discussed at our next Procurement/ Market Access Working Party in September.
